Description
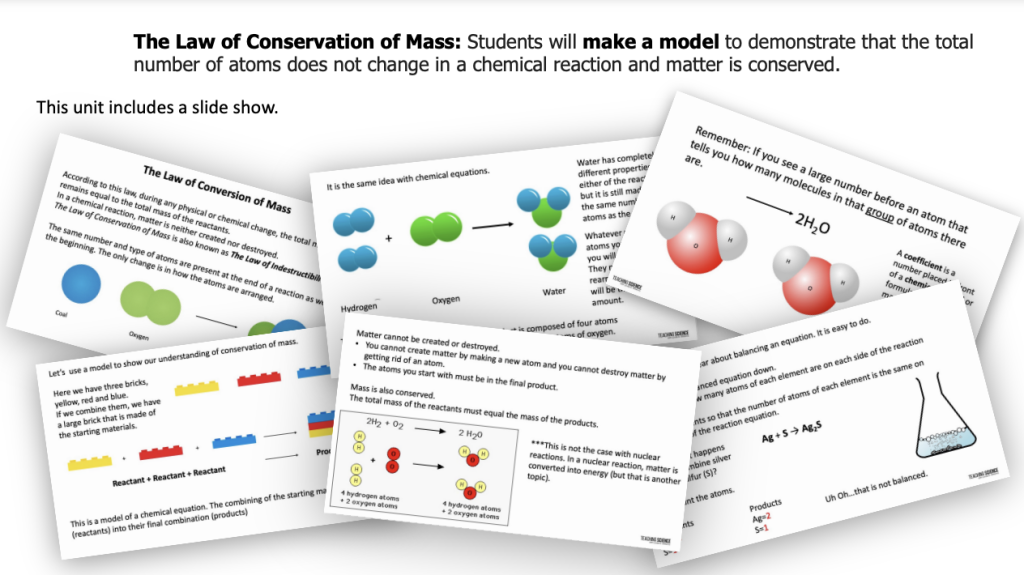
Do you need to teach about the Law of Conservation of Mass? In this unit students will make a model to demonstrate that the total number of atoms does not change in a chemical reaction and matter is conserved. Students will learn about the conservation of mass and matter through hands-on science investigations, developing and using models, and follow-up pages. Students will learn to count atoms. They will also learn that the same number and type of atoms in the reactants are found in the product, although they might be rearranged.
This resource includes:
- a slide show
- strategic notetaking guide
- two labs
- a hands-on activity making a physical model
- an informational text article on the conservation of mass
- response pages
- a quiz
Teachers Love This Resource!
Thomas ⭐️ ⭐️ ⭐️ ⭐️ ⭐️ Extremely Satisfied
I am so glad to have this resource that focuses on conservation without requiring students to do more advanced work, like balance equations. Activities were good, especially the hands-on one with blocks.
Michaun ⭐️ ⭐️ ⭐️ ⭐️ ⭐️ Extremely Satisfied
My students loved the labs and I found the slide show and strategic note-taking guide super helpful. This author does such a great job aligning to standards. I have purchased several of Lynda’s resources, and they are always terrific!
Science Standards Addressed in this Unit:
✅ NGSS MS-PS1-5. Develop and use a model to describe how the total number of atoms does not change in a chemical reaction and thus mass is conserved.
✅ Utah SEEd Standard 8.1.6 Develop a model to describe how the total number of atoms does not change in a chemical reaction, indicating that matter is conserved. Emphasize demonstrations of an understanding of the law of conservation of matter. Balancing equations and stoichiometry will be learned at the high school level. (PS1.B)
The focus of this unit is on the introduction to the concept of the Law of Conservation of Mass. The words: mass and matter are both used in this lesson and in the scientific community when talking about this law. Students will be introduced to balancing equations, but not required to balance equations since that is a standard for later grades.
The labs use simple materials:
- vinegar
- powdered drink mix
- baking soda
- water
- a balloon
- and a scale that can measure in grams
- You will also need some plastic building blocks for the hands-on physical model.

Want to read more about this resource? Check out my blog post here!
Save money and purchase as part of this bundle: Matter and Its Interactions









