Description
States of Matter Unit: Students will learn that applying heat and cold can change water from one state to another. Students will also learn about the characteristics of each state of matter including volume, shape, rigidness, and molecule arrangement. They will learn that some changes caused by heat can be reversed and some cannot. This resource has a hands-on 5 E inquiry-based lab and response pages. Parts of this can be used as a Distance Learning Packet.
NGSS 2-PS1-4. Construct an argument with evidence that some changes caused by heating or cooling can be reversed and some cannot.
This resource also aligns with Utah SEEd Standard 2.3.4 Obtain, evaluate, and communicate information about changes in matter caused by heating or cooling. Emphasize that some changes can be reversed and some cannot. Examples of reversible changes could include freezing water or melting crayons. Examples of irreversible changes could include cooking an egg or burning wood. (PS1.B)
and NJSLS • 2-PS1-4 Construct an argument with evidence that some changes caused by heating or cooling can be reversed and some cannot
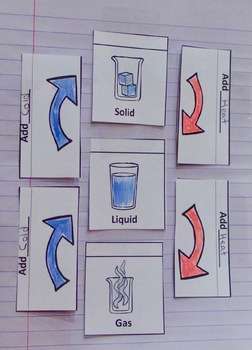
This resource focuses more on basic science concepts of three main states of matter, and how adding heat or cold to them can change them into another state of matter. This resource also looks at the properties of each state of matter (gas has an unfixed shape and takes on the shape of the container). See more of this unit here: States of Matter
Science Concepts in this Resource:
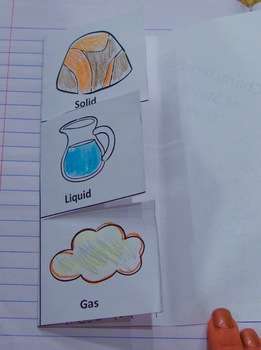
Solids have a fixed volume and shape
Liquids take on the shape of the container
Gas takes on the shape and volume of the container
Molecules in solids are close together and vibrate slightly
Molecules in liquid move around and are farther apart
Molecules in gas sip around quickly are ar spread out
States can be changed by adding heat or cold